  
Here with the fact that oxygen exists in nature as a diatomic molecule, O 2. Notice also that the example deals with atoms of oxygen we are not concerned In this example, notice that each factor involving a mole states the chemicalĬomposition of the mole: "1 mol CO 2" and "2 mol O atoms." As problemsīecome increasingly complex, this bookkeeping habit becomes especially important. How many atoms of oxygen are in 0.262 g carbon dioxide, CO 2? Used as a conversion factor in solving problems. This relationship between mass and moles of a compound is often Note that these individual masses add up to our calculated formula weightįor the acid. For example, one mole of sulfuric acid contains:Ģ mol hydrogen atoms, weighing 2(1.008 g) or 2.016 gġ mol sulfur atoms, weighing 1(32.07 g) or 32.07 gĤ mol oxygen atoms, weighing 4(16.00 g) or 64.00 g Of a compound also tells how many moles of a particular element are contained 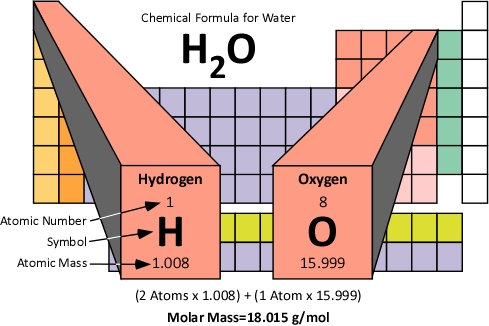
Thus it follows that one mole of sulfuric acid has a mass of 98.09 g. InĮxample 6.8 we calculated the formula weight of sulfuric acid to be 98.09 amu, Have a mass in grams equal to the formula weight of that compound or ion. We can also have moles of compounds or of ions. In that section, we talked about moles of atomsĪnd you learned that one mole of a particular element has a mass in grams equal With each element's atomic weight being multiplied by the number of atoms ofĪs 6.02 X 10 23 items. Ion is the sum of the atomic weights of all elements in the compound or ion, The formula weight (sometimes called the molecular weight) of a compound or 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
